Resources
Insights
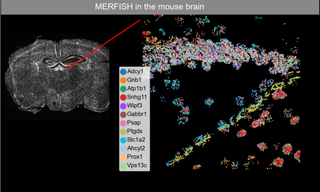
White Paper: Laser Light Engine Output Stability
This paper focusses on the temporal characteristics of CELESTA light output, presenting typical data over different timescales and using a variety of photometric detectors.
White Paper
Spotlights
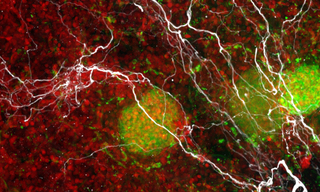
Case Study: A Symphony of Discovery- VOLTA and Pharmacological Cardiac Safety Profiling
Case Study
Press Release

Lumencor Opens New Sales and Demo Center at U-PARC, University of Pittsburgh
Insights

Elevating Confocal Imaging: ZIVA Light Engine for Yokogawa CSU
Video
Spotlights
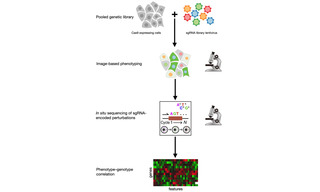
Optical Pooled Screening using the CELESTA Light Engine
Case Study
Insights

Laser Light Engine Stability
Video
Insights
Case Study: ZIVA Light Engine for Yokogawa CSU
Insights
Introducing a Game-Changing New ZIVA Light Engine
Insights
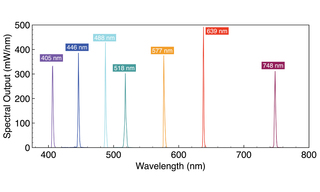
Advances in Laser Illumination for Spinning Disk Confocal Microscopy
Case Study
Insights

Introducing the VOLTA Scanner: An Optical High-Throughput Plate Reader for Transmembrane Ion-Channel Analyses
Video
Insights

Nikon BioImaging Centers depend on Lumencor for Illumination
Case Study
Spotlights

How will the FDA's Modernization Act affect you?
Insights
Lumencor Light Engines for Microscopy at VT’s Neuroscience Centers
Case Study
Insights

Oregon’s Willamette University Graduate-Level Research by Undergraduates, using Lumencor Light Engines
Case Study