Neurodevelopment in Schizophrenia poses major challenges for experimental study at least in part due to genetic complexity, cellular diversity, and limitations in accessing living human brain tissue. To overcome such barriers researchers often use two complementary human stem cell–derived models: adherent cortical organoids and NGN2 (Neurogenin-2) induced neurons. Adherent cortical organoids form three-dimensional cultures containing diverse cortical neuron types, enabling analysis of network development and long-term maturation over months [1]. In contrast, NGN2 neurons generate rapid, two-dimensional, homogeneous populations of excitatory neurons that display robust activity within weeks, making them well suited for scalable, functional assays and high-throughput screening [2].
With the added insight that stem cell models offer a window into the neural development of the schizophrenic brain, the quantification of patient-derived neurons’ collective function is a priority [3]. At Columbia University’s Mortimer B. Zuckerman Mind Brain and Behavior Institute, researchers use NGN2-neurons, yielding reproducible populations of excitatory cortical neurons that scale reliably across experiments. Calcium imaging provides a powerful functional readout in these NGN2-neuron networks. When a neuron fires an action potential, voltage-gated calcium channels open and intracellular calcium rises sharply [4]. Fluorescence calcium indicators convert transient, ionic changes into fluorescence emission, that can be quantitatively detected via light microscopy across thousands of cells simultaneously. Coupling calcium-sensitive reporters with high-speed optical microscopy enables noninvasive, population-level measurement of neural activity, synchrony, and network dynamics. Interpreting these rich image sequences requires sophisticated theoretical and numerical approaches [5].
Building directly on the ability to measure neural activity with calcium imaging at scale, Lumencor’s TARGA Imager represents a transformative step in the development of optical imaging hardware for the study of neurodevelopmental conditions such as schizophrenia. Well suited to workflows where NGN2-neuron cultures are studied across multiple conditions in parallel (Figure 1), TARGA delivers calcium fluorescence images over millimeter-scale fields of view within standard 96-well plates, entire well areas, while maintaining high-speed, faster-than video rate imaging with precision resolution. These capabilities allow researchers to observe chemical communications across large neuronal networks rather than isolated cells in real time. Images can be acquired at frequencies up to 100 Hz, enabling capture of fast calcium transients of collective neuronal dynamics. Rapid switching of multicolor excitation light supports
multiplexed fluorescence dyes, linking functional activity with
cellular structure and organization.
Overall, TARGA achieves high spatial, temporal, and spectral resolution simultaneously with precise, automated opto-mechanical architecture. This data is well matched to modern image analysis algorithms, generating robust fluorescence traces from complex neuronal populations (Figure 2). In combination, these features make the TARGA Imager a revelatory neuroscience tool, uniquely enabling visualization of emergent collective behavior at millimeter scale with exceptional resolution. Such integrated performance accelerates discovery by bridging cellular mechanisms and systems-level phenotypes relevant to schizophrenia pathophysiology and therapeutic screening. By uniting scale, speed, and precision in a single optical platform, TARGA empowers researchers to probe experimental neuroscience and strengthens translational studies of complex psychiatric disease at population scale.
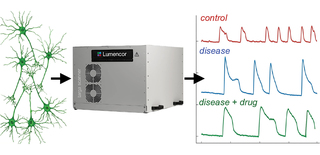
Figure 1: TARGA implementation for NGN2-neurons calcium imaging workflow
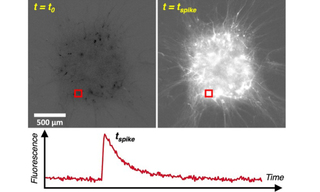
Figure 2: Calcium fluorescence trace detection from millimeter scale neuronal tissue
- May 06, 2026
- [1] Van der Kroeg et al. Human adherent cortical organoids in a multiwell format. eLife. 2024. 13:e98340.(opens in new window)
- [2] Shan et al. Fully defined NGN2 neuron protocol reveals diverse signatures of neuronal maturation. Cell Reports Methods. 2024. 4:100858.(opens in new window)
- [3] Rao et al. Aberrant pace of cortical neuron development in brain organoids from patients with 22q11.2 deletion syndrome‑associated schizophrenia. Nature Communications. 2025. 16:6986.(opens in new window)
- [4] Zhang et al. Fast and sensitive GCaMP calcium indicators for imaging neural populations. Nature. 2023. 615:884–891.(opens in new window)
- [5] . Pasarkar et al. maskNMF: A denoise‑sparsen‑detect approach for extracting neural signals from dense imaging data. bioRxiv. 2023. 2023.09.14.557777.(opens in new window)


