Diagnostic Testing
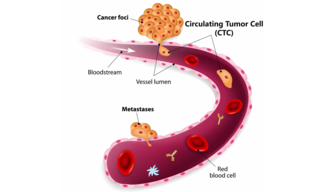
Fluorescence detection powered by Lumencor’s solid-state light sources is used in many diagnostic testing applications including circulating tumor cell (CTC) detection, immunofluorescence assays and fluorescence in situ hybridization (FISH) analysis. Furthermore gene expression analysis is increasingly evolving from a research technique to platform technology for diagnostic testing. As in other application areas, producing optimized microscope illumination for fluorescence detection demands detailed attention to the spatial, spectral and temporal characteristics of light. In diagnostic testing, regulatory requirements introduce an additional layer of complexity. Lumencor is well equipped, in terms of experience and engineering capabilities, to meet these challenges. Our Beaverton, OR manufacturing facility operates under an ISO 13485:2016 medical devices quality management system and we maintain an in-house certification and testing laboratory.
Commonly Used Products
Diagnostic Testing